Antibodies that bind to preselected regions of the urokinase molecule have been developed. These antibodies can be used to measure small quantities of each of three molecular forms of urokinase that could be contained in microsamples or conditioned media harvested from cultures of mammalian cells. Previously available antibodies and assay techniques do not yield both clear distinctions among, and measurements of, all three forms.
Urokinase is a zymogen that is synthesized in a single-chain form, called “ScuPA,” which is composed of 411 amino acid residues (see figure). ScuPA has very little enzyme activity, but it can be activated in two ways: (1) by cleavage of the peptide bond lysine 158/isoleucine 159 and the loss of lysine 158 to obtain the high molecular- weight (HMW) form of the enzyme or (2) by cleavage of the bond lysine 135/lysine 136 to obtain the low-molecular- weight (LMW) form of the enzyme.
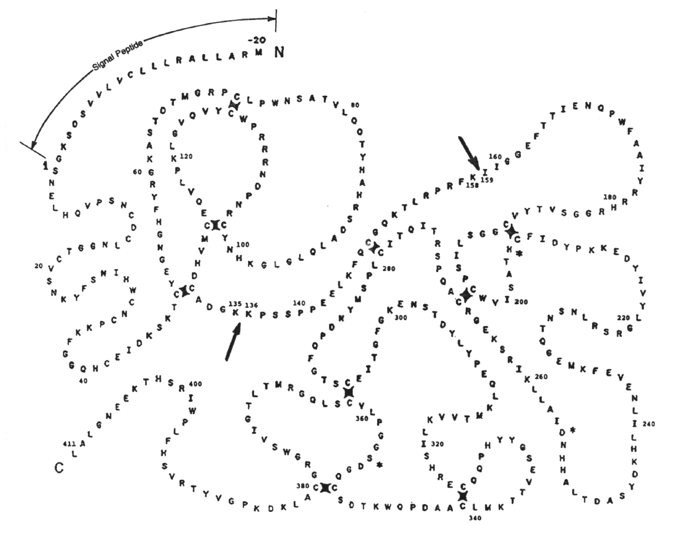
The antibodies in question were produced in mice and rabbits by use of peptides as immunogens. The peptides were selected to obtain antibodies that bind to regions of ScuPA that include the lysine 158/isoleucine 159 and the lysine 135/lysine 136 bonds. The antibodies include monoclonal and polyclonal ones that yield indications as to whether either of these bonds is intact. The polyclonal antibodies include ones that preferentially bind to the HMW or LMW forms of the urokinase molecule. The monoclonal antibodies include ones that discriminate between the ScuPA and the HMW form. A combination of these molecular-specific antibodies will enable simultaneous assays of the ScuPA, HMW, and LMW forms in the same specimen of culture medium.
This work was done by Dennis R. Morrison of Johnson Space Center and M. Zouhair Atassi of Baylor College of Medicine. For further information, contact the Johnson Commercial Technology Office at (281) 483-3809.
This invention is owned by NASA, and a patent application has been filed. Inquiries concerning nonexclusive or exclusive license for its commercial development should be addressed to
the Patent Counsel
Johnson Space Center
(281) 483-0837.
Refer to MSC-21947.

