A method of growing bioengineered tissues for use in surgical replacement of damaged anterior cruciate ligaments has been invented. An anterior cruciate ligament is one of two ligaments (the other being the posterior cruciate ligament) that cross in the middle of a knee joint and act to prevent the bones in the knee from sliding forward and backward relative to each other. Anterior cruciate ligaments are frequently torn in sports injuries and traffic accidents, resulting in pain and severe limitations on mobility. By making it possible to grow replacement anterior cruciate ligaments that structurally and functionally resemble natural ones more closely than do totally synthetic replacements, the method could create new opportunities for full or nearly full restoration of functionality in injured knees. The method is also adaptable to the growth of bioengineered replacements for other ligaments (e.g., other knee ligaments as well as those in the hands, wrists, and elbows) and to the production of tissues other than ligaments, including cartilage, bones, muscles, and blood vessels.
The method is based on the finding that the histomorphological properties of a bioengineered tissue grown in vitro from pluripotent cells within a matrix are affected by the direct application of mechanical force to the matrix during growth generation. This finding provides important new insights into the relationships among mechanical stress, biochemical and cell-immobilization methods, and cell differentiation, and is applicable to the production of the variety of tissues mentioned above. Moreover, this finding can be generalized to non-mechanical (e.g., chemical and electromagnetic) stimuli that are experienced in vivo by tissues of interest and, hence, the method can be modified to incorporate such stimuli in the ex vivo growth of replacements for the various tissues mentioned above.
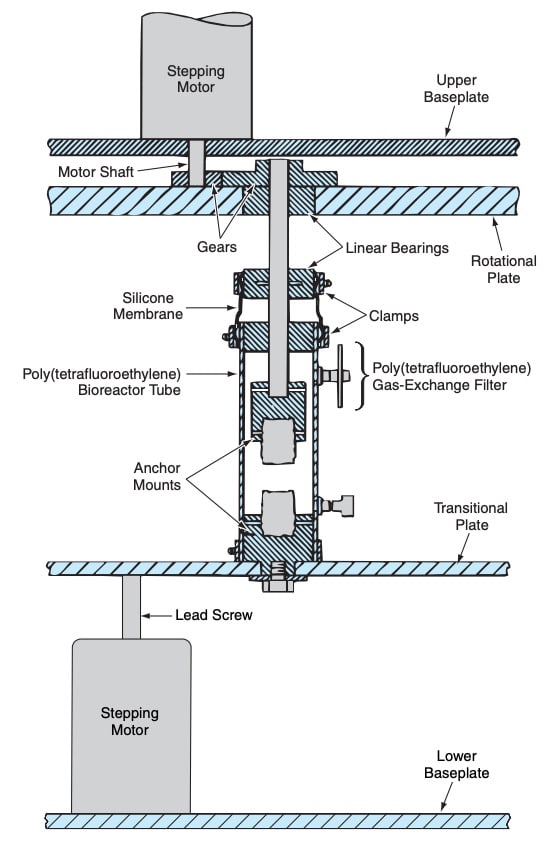
In this method, a three-dimensional matrix made of a suitable material is seeded with pluripotent stem cells. The patient’s bone-marrow stromal cells are preferably used as the pluripotent cells in this method. Suitable matrix materials are materials to which cells can adhere — for example, collagen type I. The seeded matrix is attached to anchors at opposite ends and then the cells in the matrix are cultured under conditions appropriate for the growth and regeneration of cells. Suitable anchor materials are materials to which the matrix can attach; examples include demineralized bone and Goinopra coral that has been treated to convert its calcium carbonate to calcium phosphate.
During the growth process, the matrix is subjected to a combination of tension, compression, torsion, and shear stresses via movement of one or both of the anchors. The figure depicts the combination of (1) a bioreactor tube within which the growth process takes place and (2) an apparatus that operates under computer control to generate the required motions. Optimally, the stresses should mimic those to which the anterior cruciate ligament is subjected in vivo during normal activity. The bioengineered ligament produced by this method is characterized by a cellular orientation and/or matrix crimp pattern in the direction of the applied mechanical forces, and by the production of collagen type I, collagen type III, and fibronectin proteins along the axis of mechanical loading. Optimally, the ligament thus produced contains fiber bundles arranged in a helical pattern.
This work was done by Gregory Altman, David L. Kaplan, Ivan Martin, and Gordana Vunjak-Novakovic of Massachusetts Institute of Technology for Johnson Space Center.
In accordance with Public Law 96-517, the contractor has elected to retain title to this invention. Inquiries concerning rights for its commercial use should be addressed to:
Technology Licensing Office
Massachusetts Institute of Technology
Five Cambridge Center, Kendall Square
Room NE25-230
Cambridge, MA 02142-1493
Phone: (617) 253-6966
Fax: (617) 258-6790
E-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Refer to MSC-23351, volume and number of this NASA Tech Briefs issue, and the page number.

