Pellets made of a high-surface-area composite of silica and titania have shown promise as means of removing elemental mercury from flue gases. With further technical development and commercialization, this material could become economically attractive as a more-effective, less-expensive alternative to activated carbons for removing mercury from exhaust streams of coal-burning power plants, which are the sources of more than 90 percent of all anthropogenic airborne mercury.
The silica/titania composite is made from a silica precursor and titania, starting with a sol-gel process in which water, a solvent, and acids are used to promote the hydrolysis and condensation reactions. After gelation, the reaction mixtures are aged, rinsed, and then dried in a series of heat treatments. The resulting composite pellets are about 5 mm long and 3 mm wide. Each pellet comprises TiO2 nanoparticles distributed in a porous SiO2 matrix. The pores are of the order of 15 nm wide. The pellets are characterized by specific surface area of about 300 m2/g.
This composite material removes elemental mercury from air or a flue-gas mixture through a synergistic combination of (1) adsorption on the surfaces of the composite pellets and (2) photocatalytic oxidation in the presence of ultraviolet light. The mercury oxide remains on the surfaces of the pellets as a solid deposit. From time to time, the mercury oxide can be extracted from the pellets — thereby both regenerating the pellets and making the mercury available for recycling — for example, by rinsing the pellets with acid.
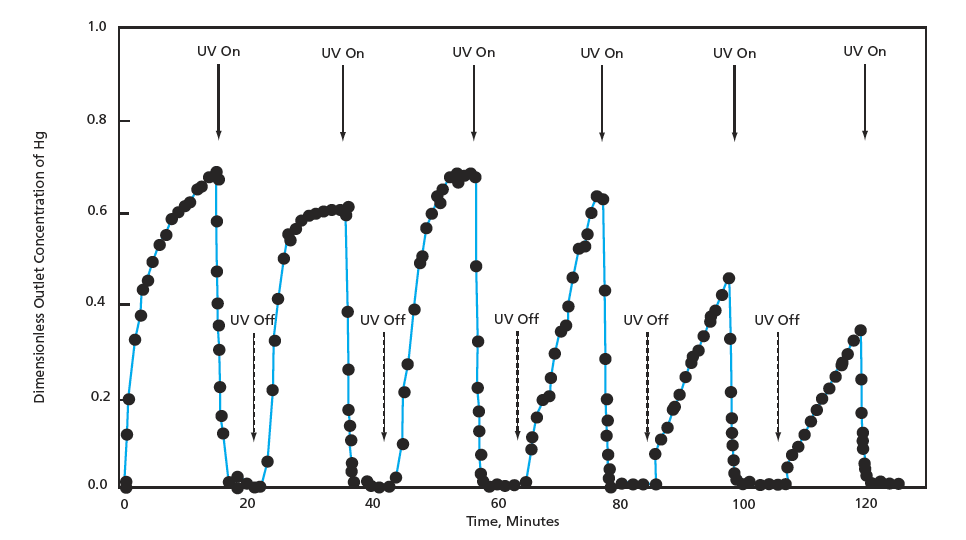
The adsorptive capacity of the composite material is great enough to enable continuous concentration of mercury onto the pellets without continuous use of ultraviolet light (see figure). Intermittent ultraviolet irradiation suffices to ensure oxidation of adsorbed mercury and mercury vapor and to regenerate the adsorbent. The efficiency of removal of Hg can easily be made as high as 99 percent or greater while ultraviolet light is on. Moreover, the photocatalytic oxidation can be said to activate the adsorbent in that it increases the subsequent adsorption capacity in the absence of ultraviolet light.
The mercury-adsorption capacity of the pellets is high although at the 3-mm pellet width, only a thin outer layer is utilized effectively. A TiO2 loading of 13 weight percent has been found to result in the best removal of Hg, both with and without ultraviolet light. Humidity has been found to impede adsorption, thereby reducing the overall Hg-removal efficiency. An examination of the effects of flow velocity revealed that adsorption is the rate-limiting step, suggesting a need to improve mass-transfer characteristics to obtain better performance.
This work was done by David Mazyck, Danielle Londeree, Chang-Yu Wu, Kevin Powers, and Erik Pitoniak of the University of Florida for Johnson Space Center.
In accordance with Public Law 96-517, the contractor has elected to retain title to this invention. Inquiries concerning rights for its commercial use should be addressed to:
University of Florida,
Environmental Engineering
306 Black Hall
Gainesville, FL 32611
Refer to MSC-23624, volume and number of this NASA Tech Briefs issue, and the page number.

