The inherently interdisciplinary nature of developing instrumentation for life sciences requires a high level of collaboration between scientists and engineers across the fields of analytical or clinical chemistry, optics, mechanics, material science, and microbiology. Moreover, product development teams are competing for first-to-market benefits that are driven by intellectual property lifetimes and insuring an installed base quickly to realize recurring consumable sales. Concurrently, product designers need to comply with current Good Manufacturing Practices (cGMP). System-level modeling enables adherence to the methodical design process without the cost and time associated with iterative hardware prototyping and laboratory and clinical testing.
Fluorophore Beginnings


Based on the target protein, platform development began with the selection of fluorescent dye, Alexa Fluor 488. The dye was chosen because it has the proper reactive groups and accommodates assay conditions including photostability and pH. Optical properties of the dye, including relative excitation and emission curves and the peak molar extinction coefficient, are retrieved from the Invitrogen Molecular Probe Products database resident in TracePro and TracePro Bridge for SolidWorks.
The dye was modeled in an aqueous solution with a pH>8. Quantum efficiency was determined by biochemists to be 0.92 as a free dye and 0.55 when conjugated to the target protein. The initial design tests the feasibility of the fluorophore concentration at 10 E -10 moles per liter. A range of concentrations is modeled to validate adherence to the dynamic range specification.
Source and Detector Models
Based on the peak excitation wavelength of Alexa Fluor 488 dye, an excitation source is selected that balances a wavelength closest to peak excitation, luminous flux, and cost. A blue LED, Kingbright (City of Industry, CA) model WP7524PBC/J, with wavelength of 467 nm, ±22 nm, was selected and its SolidWorks model and optical properties were downloaded from the Kingbright Web site and imported into the TracePro model. Luminous flux and directionality of the LED’s output were characterized in lab and imported into the TracePro model.
Based on the peak emission wavelength of the Alexa Fluor 488 dye (517 nm), a detector was selected that balanced cost and responsivity. A silicon photodiode was selected and its spectral responsivity curve was imported into the TracePro model (see Figure 2).
Fluorescence Filtering
Filter selection is most often a complex analysis of the spectral relationships of fluorophores, optical filters, excitation sources, and detectors. Because off-the-shelf components offer several ways to minimize costs in product development, purchasing, manufacturing, quality, and reliability within the DFM process, off-the-shelf filters available from commercial suppliers such as Newport Corporation (Irvine, CA) and Omega Optical (Brattleboro, VT) were considered and modeled. Optical and mechanical characteristics of these off-the-shelf filters were imported from TracePro’s libraries and modeled in the system. Narrowband excitation and emission filters centered on the Alexa Fluor 488 dye’s absorption and emission peaks were selected to minimize spectral overlap of the emission signal with the excitation signal and increase signal isolation.
Filter selection was further complicated by fluorophores’ significantly different spectral properties in a particular application such as nucleic acid stains bound to RNA compared to fluorophores in aqueous solution. Complex and application-specific characteristics of the fluorophores’ sample preparation and biological target were then modeled in combination with the selected filter pair by iterating and analyzing quantum efficiency and fluorophore concentration values directly in the TracePro and SolidWorks model.
Simulation and Analysis
The complete opto-mechanical system, including the fluorescence-tagged biological sample, was modeled and documented in SolidWorks with the TracePro Bridge add-in. The single, archived SolidWorks file was then opened with TracePro for optical simulation and analysis. Source rays propagate through the model with portions of the flux of each ray allocated to absorption, specular reflection and transmission, fluorescence, polarization, and scattering.
From the simulation, contributions to sensitivity were analyzed, including system throughput at the detector, flux absorbed by surfaces and bulk material, and stray and scattered light from mechanical and optical surfaces. Contributions to dynamic range were analyzed by changing the concentration and quantum efficiency of the dye to simulate the breadth of addressable sample preparations.
Based on the TracePro irradiance map, designers concluded that the sensitivity specification was not achieved. From the TracePro flux report, it was concluded that there were insufficient fluorescence emission photons captured, and unwanted stray light from optical and mechanical components reached the detector.
Design for Manufacturability
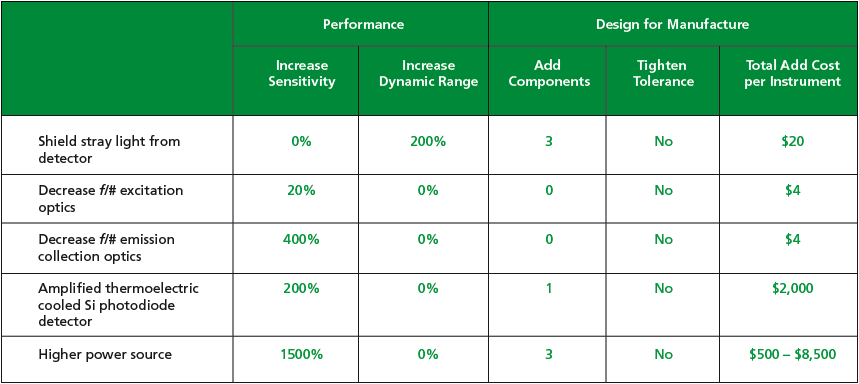
Component-level improvements affecting system level sensitivity were identified relative to driving DFM factors such as component cost, number of components, and manufacturing tolerances (see Figure 3).
From the TracePro and DFM analyses, the design team concluded that the sensitivity specification may be achieved by increasing the f/# on both the LED and detector collection optics and modifying the mechanics to reduce stray light at the detector. These design modifications offered the largest improvement in performance at orders of magnitude less expense.
The design of the new collection lenses was realized and toleranced in commercially available lens design software and translated to SolidWorks with LensWorks into the single archived SolidWorks file. Mechanics were modified and a single set of documentation was updated in SolidWorks. The same SolidWorks file was then opened by TracePro for design validation.
The development of the fluorescence spectroscopy product platform demonstrates that with an integrated set of software design tools, a disciplined DFM product development process can be executed effectively and efficiently within a multidisciplinary design team. System-level performance and component-level specifications can be communicated across technical disciplines while data and design integrity can be insured by documenting in compliance with cGMP. Product cost and time to market are minimized with improved product quality and reliability.
This article was written by Eric Heinz of Heinz Optical Engineering Co. (San Diego, CA), and Edward Freniere, Ph.D., Richard Hassler; and Linda Smith from Lambda Research Corporation (Littleton, MA), with acknowledgement to Iain Johnson at Invitrogen Corporation (Carlsbad, CA). For more information, contact Ms. Smith at

