By capping liquids with graphene (an ultrathin sheet of pure carbon), researchers can easily image and analyze liquid interfaces and the surface of nanometer-scale objects immersed in liquids. In the imaging technique known as photoemission electron microscopy (PEEM), ultraviolet light or X-rays bombard a sample, stimulating the material to release electrons from a region at or just beneath its surface. Electric fields act as lenses, focusing the emitted electrons to create an image.

The method has been used for decades to discern such fine-scale features as the patterns of chemical reactions on the surface of catalysts, the magnetic field structure of memory devices, and the molecular architecture of biological compounds. PEEM has typically been restricted to solid surfaces that are in a high vacuum environment; the method hasn’t had the ability to study liquids and gases at ordinary pressures. A liquid sample, for instance, would evaporate and create sparks if directly exposed to the high vacuum in the PEEM setup.
In the past, scientists have attempted to overcome these challenges by using a technique known as differential pumping that bridges the gap between the high pressure of the sample and the essentially zero pressure of the microscope. But such instrumentation is not sufficient to reach truly ambient pressure conditions, and is too expensive and not widely accessible for routine use. Sealing a liquid or gaseous sample with a graphene lid just one or two atomic layers in thickness keeps the sample at atmospheric pressure while allowing the system to be placed under vacuum.
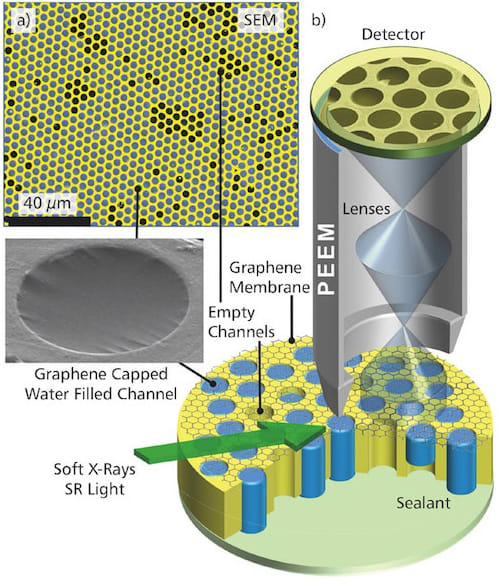
The graphene lid enables electrons emitted by the test liquid to pass nearly unimpeded to the detector, yet keeps the liquid from escaping into the vacuum of the PEEM. An array of the lids retained the liquid samples for hours under high vacuum — long enough to perform routine electron imaging and spectroscopy experiments. The lids allow the liquid to be changed while an experiment is in progress, helping researchers understand the behavior of the sample under different chemical environments.
In addition, because the setup uses an array of identical lids, each can be a different sample, and the technique can be used in conjunction with powerful statistical analysis, data mining, and pattern recognition methods.
For more information, contact Ben Stein at

