Metallurgists have typically made a metal harder by bending it, twisting it, and smashing it with a hammer. The idea being: The smaller the metal's grains, the stronger the structure.
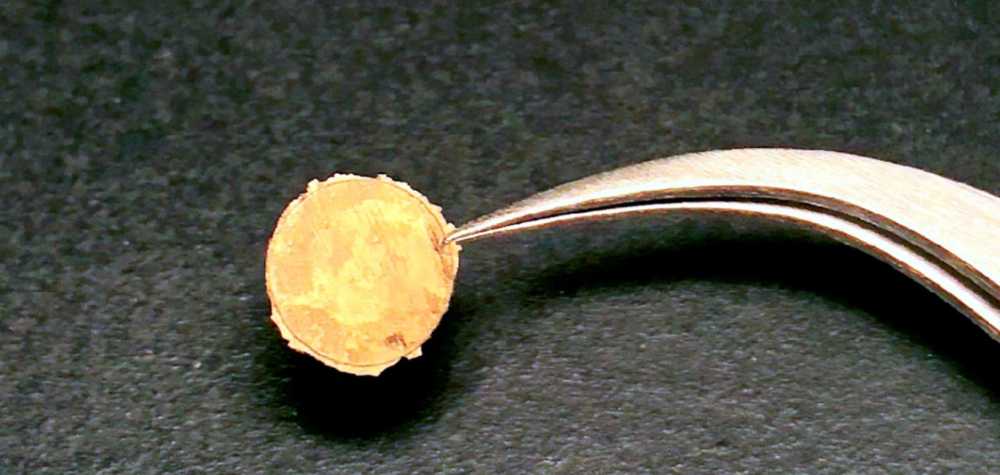
Brown University researchers have discovered a reverse approach to strengthening metal: squeezing the tiny metal nanoparticles together. The resulting "coin" enables precise customization and control of the metal's microstructures.
In a paper published in the journal Chem , the scientists demonstrated a way of smashing individual metal nanoclusters together to form solid macro-scale hunks of solid metal.
Tests revealed that the resulting metals measured up to four times harder than naturally occurring metal structures. Other properties like electrical conduction and light reflectance were virtually identical to standard metals.
The Brown University researchers, led by assistant professor of chemistry Dr. Ou Chen, used a homemade pressure piston cell to apply the pressure — about 0.6 gigapascals (GPa) — to consolidate the nanoparticle powders and form free-standing coins.
"It does look like a small coin in your wallet but made from nanocrystals," Chen told Tech Briefs in the short Q&A below.
The centimeter-scale coins were made from nanoparticles of gold, silver, palladium and other metals.
Items of this size could be useful for making high-performance coating materials, electrodes, or thermoelectric generators, says Chen.
The nanoparticles could also potentially be used as a kind of "ink" over other existing materials. Once pressed and consolidated, the nanoparticles could form a strong metal coating.
“What we’ve done is create nanoparticle building blocks that fuse together when you squeeze them," said Chen in a university press release. "This way we can have uniform grain sizes that can be precisely tuned for enhanced properties.”
To create the coins, the researchers had to apply a chemical treatment on the nanoparticles.
Metal nanoparticles are typically covered with organic molecules called ligands, which generally prevent the formation of metal-metal bonds between particles. Chen and his team found a way to strip those ligands away chemically, allowing the nanoparticles to fuse together with just a bit of pressure.
The process could easily be scaled up to make super-hard metal coatings or larger industrial components, says Chen, and potentially used on any kind of metal.
Chen and the team even created a metallic glass — a material that lacks the regularly repeating crystalline structure of normal metal — with amorphous palladium nanoparticles.
Chen says he’s hopeful that the technique could one day be widely used for commercial products. The patented chemical treatment used on the nanoclusters is fairly simple, and the pressures used to squeeze them together are well within the range of standard industrial equipment.
In an edited interview with Tech Briefs below, Dr. Chen explains why he thinks the manufacturing method has so much potential for industry and scientific research.
Tech Briefs: What inspired the idea to smash the tiny nanoparticles together to make super-hard metals?
Dr. Ou Chen: People know that metalsmiths harden metals in ways such as "strike while the iron is hot." The science behind it is to break down crystal domains and manipulate the grain-boundary condition inside metals. In my lab, we have been working on nanomaterials and high-pressure chemistry for years. Inspired by these traditional methods and ideas, we realized that we may be able to engineer the grain-boundary conditions in a much more controllable way: fusing nanoparticles together to make grain-boundary designable metals.
Tech Briefs: Does it look like a coin? And is the idea that these coins can be smashed together to make a coating that can then be put over an existing metal to make it harder?
Dr. Ou Chen: We showed several photos of the "coins" we made in the article published on Chem. Yes, it does look like a small coin in your wallet but made from nanocrystals. One of the advantages of colloidal nanoparticles is that these tiny particles can disperse in different solutions which can be further processed for applications such as printing or coating. So, in principle, yes, we believe our method can be used to coat and strengthen existing metals or other materials.
Tech Briefs: Does the making of these coins require a big, complicated setup?
Dr. Ou Chen: Actually, unlike many other high pressure chemistry studies, our "coining" method does not need a complicated setup. Once we had our nanoparticles ready, we used our small homemade piston pressure cell to press the nanoparticles and make these coins. This simple setup requirement makes the method easy scaling up and adaptable for different applications.
Tech Briefs: Where will these kinds of metals be used? What needs to happen before this process can be used in commercial products?
Dr. Ou Chen: We believe there will be many application outlets for these metals and the coining method, such as superhard coatings, supporting materials, and high performance electrodes.
There is still a lot more that needs to be studied, such as the relationship between materials properties and detailed nanosized grain-boundary conditions. I am sure we and other researchers will continue working along the direction. I do hope to see commercial products produced from our method in the near future.
Chen’s coauthors on the paper were Yasutaka Nagaoka, Masayuki Suda, Insun Yoon, Na Chen, Hanjun Yang, Yuzi Liu, Brendan A. Anzures, Stephen W. Parman, Zhongwu Wang, Michael Grünwald and Hiroshi M. Yamamoto. The research was supported by the National Science Foundation (CMMI-1934314, DMR-1332208, DMR-1848499) and the U.S. Department of Energy (DE-AC02-06CH11357).
What do you think? Share your questions and comments below
Transcript
00:00:07 Steel and plastic. These two materials are essential to so much of our infrastructure and technology, and they have a complementary set of strengths and weaknesses. Steel is strong and hard, but difficult to shape intricately. While plastic can take on just about any form, it's weak and soft. So wouldn't it be nice if there were one material as strong as the strongest steel and as shapeable as plastic?
00:00:34 Well, a lot of scientists and technologists are getting excited about a relatively recent invention called metallic glass with both of those properties, and more. Metallic glasses look shiny and opaque, like metals, and also like metals, they conduct heat and electricity. But they're way stronger than most metals, which means they can withstand a lot of force without getting bent or dented, making ultrasharp scalpels, and ultrastrong electronics cases,
00:01:02 hinges, screws; the list goes on. Metallic glasses also have an incredible ability to store and release elastic energy, which makes them perfect for sports equipment, like tennis racquets, golf clubs, and skis. They're resistant to corrosion,
00:01:18 and can be cast into complex shapes with mirror-like surfaces in a single molding step. Despite their strength at room temperature, if you go up a few hundred degrees Celsius, they soften significantly, and can be deformed into any shape you like. Cool them back down, and they regain the strength. So where do all of these wondrous attributes come from? In essence, they have to do with metallic glass' unique atomic structure.
00:01:46 Most metals are crystalline as solids. That means that if you zoomed in close enough to see the individual atoms, they'd be neatly lined up in an orderly, repeating pattern that extends throughout the whole material. Ice is crystalline, and so are diamonds, and salt. If you heat these materials up enough and melt them, the atoms can jiggle freely and move randomly, but when you cool them back down,
00:02:10 the atoms reorganize themselves, reestablishing the crystal. But what if you could cool a molten metal so fast that the atoms couldn't find their places again, so that the material was solid, but with the chaotic, amorphous internal structure of a liquid? That's metallic glass. This structure has the added benefit of lacking the grain boundaries that most metals have. Those are weak spots where the material is more susceptible to scratches
00:02:37 or corrosion. The first metallic glass was made in 1960 from gold and silicon. It wasn't easy to make. Because metal atoms crystallize so rapidly, scientists had to cool the alloy down incredibly fast, a million degrees Kelvin per second, by shooting tiny droplets at cold copper plates, or spinning ultrathin ribbons. At that time, metallic glasses could only be tens or hundreds of microns thick, which was too thin for most practical applications.
00:03:09 But since then, scientists have figured out that if you blend several metals that mix with each other freely, but can't easily crystallize together, usually because they have very different atomic sizes, the mixture crystallizes much more slowly. That means you don't have to cool it down as fast, so the material can be thicker, centimeters instead of micrometers. These materials are called bulk metallic glasses, or BMGs. Now there are hundreds of different BMGs,
00:03:37 so why aren't all of our bridges and cars made out of them? Many of the BMGs currently available are made from expensive metals, like palladium and zirconium, and they have to be really pure because any impurities can cause crystallization. So a BMG skyscraper or space shuttle would be astronomically expensive. And despite their strength, they're not yet tough enough for load-bearing applications. When the stresses get high, they can fracture without warning, which isn't ideal for, say, a bridge.
00:04:08 But when engineers figure out how to make BMGs from cheaper metals, and how to make them even tougher, for these super materials, the sky's the limit.