
Since the inception of integrated circuits (IC) and their rapid proliferation, Moore’s Law has led to increasingly powerful ICs while also enabling miniaturization. As the battery size decreases, the relative weight proportion of the packaging envelope increases due to surface-area-to-volume factors requiring innovative means to preserve an appropriately high energy density. Thus, the twin constraints of size and the maximization of energy density leads to a complex host of engineering challenges.
At the small manufacturing scales needed for microbatteries, existing sealing technologies like thermal sealing are impractical. Currently, researchers are exploring the use of adhesives in combination with laser-cut gasket materials to deliver maximum energy density, miniaturization, and while maintaining a high degree of hermetic integrity to assure long-lived operation and efficiency.
Adhesive Specifications for Hermetic Sealing of Microbatteries
An adhesive used for the hermetic sealing of batteries must meet a host of criteria: chemical, temperature, pressure, and electrical resistance, minimal outgassing during cure, low permeability to atmospheric moisture, high adhesion to the packaging envelope materials, and appropriate modulus and thermal expansion properties. Overall, the adhesive must avoid contaminating the sensitive electrolyte solution either because of cure-byproducts or because of chemical degradation over the operating life of the battery. Further, rheology, wetting, and adhesion properties are critical for assuring a continuous, non-permeable seal.
An appropriate adhesive must have chemical resistance to the corrosive salts and the organic electrolyte present within the battery; further, during operation the cell experiences high temperatures and pressures which encourage chemical attack on the adhesive. The chemical breakdown of the adhesive poses three risks: loss of the hermetic seal with concomitant loss of the electrolyte through volatilization, the introduction of deleterious moisture to the cell internals, and the introduction of adhesive breakdown products into the battery itself.
It is then critical that the adhesive provides the utmost in chemical resistance while also engineering the battery such that the surface of the adhesive exposed to the internal electrolyte solution is minimized. Generally, to minimize exposure of the adhesive to the electrolyte, a gasket material such as Kapton or neoprene is used.1,2 Further, the adhesive must be capable of electrically isolating the cathode and anode; thus, the adhesive must withstand the chemical and electrical environment posed by the cell.
Permeability is another critical factor in providing hermeticity. The adhesive must have a low permeability to atmospheric moisture; silicone adhesives offer exceptional temperature resistance; however, they suffer from high oxygen permeability.1 Polydimethylsiloxanes (PDMS)-based adhesives are then precluded from use. Epoxy-based adhesives systems are then preferred as they offer a high degree of chemical and temperature resistance while possessing suitable permeability properties to limit the ingress of air and moisture.
To some degree, permeability depends upon the chemical structure and polymeric structure of the adhesive. However, increased cross-link density, polymer microstructure, and a glass transition temperature (Tg) will influence the degree of permeability. Further, softening above the glass transition temperature and an increase in the mobility of polymer chains at elevated temperatures may increase the permeability of the adhesive. Epoxy adhesives can be formulated to provide a high Tg, high softening point, and to possess a high temperature stability and resistance.
To maintain a hermetic seal, the adhesive must also possess properties that assure the integrity of the bond over a wide temperature range. First, the adhesive must have a high degree of adhesion with the battery casing, usually metal, and it must be applied appropriately to a cleaned surface to ensure strong adhesion and the absence of any weak boundary layers. Here, the adhesive must appropriately wet the substrate surface to form a continuous interface between the adhesive and the substrate. Further, rheological considerations such as flow and self-leveling are critical in providing a sealing layer free of defects, cracks, and channels.
As the battery may experience large temperature changes over its operating cycle, it is important to consider factors such as the coefficient of thermal expansion (CTE) of the adhesive. Stresses may accumulate at the bond line as a result of differential thermal expansion, and this may potentially lead to bond failure and a loss of hermeticity. Epoxy-based systems can be tailored to include various mineral fillers to exhibit a wide range of CTE values to minimize thermal stresses during operation.
Additional factors such as modulus, the strain response of a material under applied stress, must be balanced such that the adhesive maintains a high degree of rigidity and dimensional stability, while also yielding appropriately to dissipate accumulated stress and to resist cracking.
Processing Considerations
As the epoxy adhesive must serve as a hermetic seal and barrier to both atmospheric ingress and loss of electrolyte, the formation of a seamless interface with the metal surface to be bonded is critical. Regardless of the permeability or chemical resistance of the adhesive, if the interfacial region is not continuous, the integrity of the hermetic seal will be compromised.1 Control of the viscosity, metering, and the adhesive application method will assist with consistency in the process and in assuring seal integrity.
The rheology, viscosity, and surface tension properties of the adhesive can be tailored to improve its ability to continuously wet-out and to strongly bond to the substrate. Care should also be taken to assure that shrinkage of the epoxy or mismatch of CTE between the adhesive and substrate do not cause any gaps to form at the interface during cure or after cooldown. Shrinkage is primarily related to adhesive chemistry, but it may also be affected by the cure schedule.
Care should be extended to the mixing and degassing of the epoxy: accuracy, precision, and adherence to the manufacturer’s guidelines with respect to mix ratios is critical to assuring proper cure. Further, degassing of the epoxy prior to application is critical as micro-pockets of air or channels present within the cured epoxy will reduce the integrity of the hermetic seal. Higher cure temperatures will provide a greater degree of chemical resistance and cross-link density; this is important for minimizing leaching as well as minimizing permeability of the adhesive layer. Optimization and control of the cure temperature is then critical for assuring the integrity of the seal.
Dual-Sealing Method Using Adhesives in Combination with Polymeric Gasket
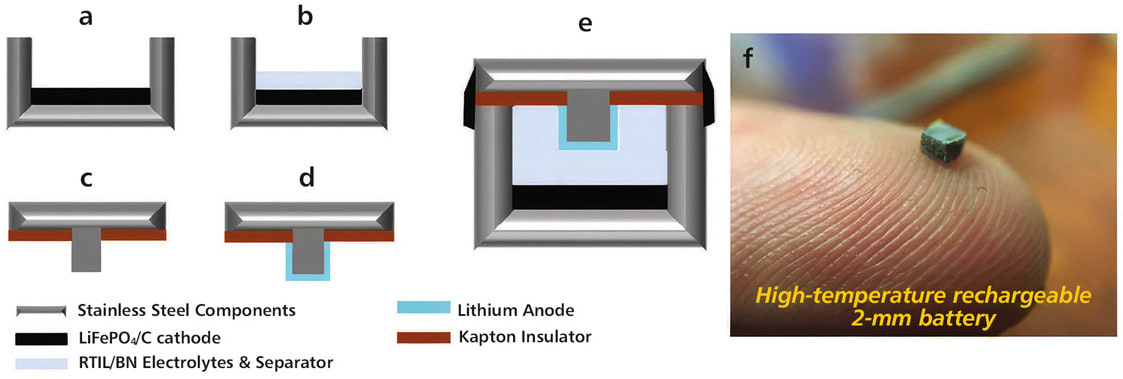
A study conducted by a team at Wayne State University reported promising results in the fabrication of a 2-mm high-temperature resistant microbattery using an epoxy that cured at room temperature in combination with a Kapton gasket/insulator.1 Their battery exhibited a discharge capacity of 62μAh at 120 °C with excellent capacity retention. A sensor duty cycle test was carried out at 120 °C, and the sensor powered by the battery provided continuous data for 22 hours; this resulting prototype also demonstrated compatibility with energy harvesting — a solar cell — to potentially lead to a miniaturized, self-directed sensing system capable of operating at high temperatures of 120 °C. Figure 1 shows a schematic representation as well as an image of the final microbattery measuring 2 mm in size.
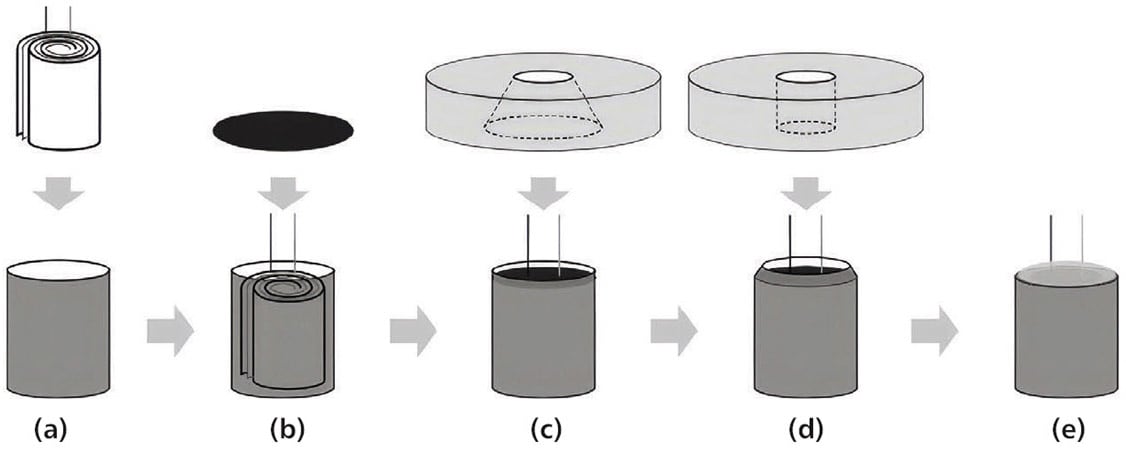
Researchers at the Pacific Northwest National Laboratory (PNNL) presented promising findings utilizing a dual-seal approach in the construction of a 3-mm wide and 6-mm long cylindrical cell microbattery.2 They utilized a combination of a neoprene gasket and a two-component epoxy in the construction of their prototype. The MB306 type microbatteries investigated in their study exhibited a discharge energy density of ~430 Wh/L and 240 Wh/kg at a discharge rate of C/7 — compared with Panasonic lithium coin cells, their prototype batteries exhibit twice the gravimetric density and similar volumetric energy density, while being only one-fifth the size. Figure 2 shows a schematic of their cylindrical microbattery.
Certifications Relevant for Industrial Adhesives
The certifications required for industrial adhesive will vary depending upon their application. As the application discussed in this article is in relation to electronic assemblies, the Restriction of Hazardous Substances (RoHS) originating from the European Union is of particular relevance. This directive seeks to restrict the use of the following chemicals in electrical and electronic products: lead (Pb), mercury (Hg), cadmium (Cd), hexavalent chromium (CrVI), polybrominated biphenyls (PBB), polybrominated diphenyl ethers (PBDE) with additional restrictions having taken effect in 2019 further restricting bis(2-etylhexyl) phthalate (DEHP), benzyl butyl phthalate (BBP), dibutyl phthalate (DBP), and diisobutyl phthalate (DIBP). All Master Bond products are RoHS 3 compliant.
Outgassing requirements, most notably NASA low outgassing requirements, seek to certify that an adhesive system will undergo minimal outgassing during the course of its operation. Outgassing may be detrimental to sensitive electronics, precision lens systems, and also the sensitive electrolyte components present within batteries. This standard originates from NASA, as exposure to the vacuums of space poses particular risk for the outgassing of volatile, migratory components trapped within adhesives or plastics. Many Master Bond products are formulated as certified to pass NASA low outgassing requirements: total mass loss (TML) of <1 percent and collected volatile condensable material (CVCM) of <0.1 percent.
This article was contributed by Master Bond (Hackensack, NJ). For more information, visit here .
REFERENCES
- Masurkar, N., Babu, G., Porchelvan, S., et al. Millimeter-scale lithium battery packaging for high-temperature sensing applications. Journal of Power Sources. 2018. Vol. 399, pp. 179-185.
- Wang, Y., Cartmell, S., Li, Q., et al. A reliable sealing method for microbatteries. Journal of Power Sources. PNNL-SA-119837. Pacific Northwest National Laboratory. 2017. Vol. 341, pp. 443-447.
- Yue, X., Grzyb, J., Padmanabha, A., Pikul, J.H. A minimal volume hermetic packaging design for high-energy-density micro-energy systems. Energies. 2020. Vol. 13, 2492.

